Abstract Today, the indirect enantiomer separation approach is the method of second choice and in most instances has been replaced by the direct approach using chiral stationary phases. Nevertheless, one should think about the underlying concept and become aware of some critical issues which are important for a general understanding of stereoselective analysis. This chapter deals with requirements, drawbacks, caveats.
LevelBasic
The utility of the indirect enantiomer separation method is connected to a number of requirements:
-
First of all, the selectand i.e. the analyte to be separated must contain a (relatively easily) derivatizable functional group such as a hydroxyl, amine, carboxylic, carbonyl group. Some common reaction pathways are outlined below. Note that the residue R1 belongs to the Chiral Derivatizing Agent (CDA) and is chiral, while the R residue in this scheme belongs to the selectand and is chiral as well.
Common Reaction Pathways in Indirect Enantiomer Separation Concepts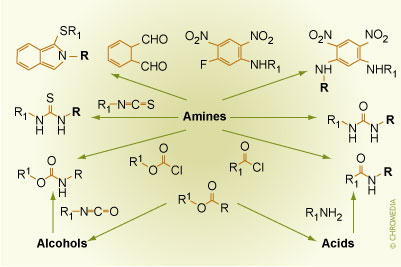 Source: Lindner (Houben-Weyl E21)
Source: Lindner (Houben-Weyl E21)
-
The chiral derivatizing agent must be highly enantiomerically pure or at least its enantiomeric impurity content must be known. Note, if the CDA contains a significant amount of enantiomeric impurity the following reactions will occur:
Indirect Approach: Equilibria in Presence of
Enantiomeric Impurity in CDA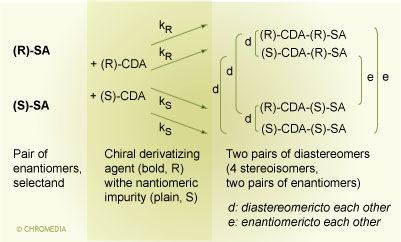
It is seen that 4 stereoisomers will be formed and if achiral chromatography e.g. RP-HPLC is employed, only two peaks will be obtained: There are two enantiomeric pairs which will be co-eluted on RP-HPLC. For example, the isomer (S)-CDA-(R)-SA, arising from the enantiomeric impurity (S)-CDA in the chiral derivatizing agent, is the enantiomer of the major isomer (R)-CDA-(S)-SA, which originates from the (S)-SA and the chiral derivatizing agent (R)-CDA. These two stereoisomers cannot be distinguished in an achiral separation system, and thus the (S)-CDA-(R)-SA and (R)-CDA-(S)-SA stereoisomers are not separated by RP-HPLC. Therefore, the quantitation of the (R)-CDA-(S)-SA would be biased by the enantiomeric impurity of the reagent. The same situation exists for the enantiomeric pair (S)-CDA-(S)-SA, arising from the enantiomeric impurity of the CDA, and (R)-CDA-(R)-SA which is the major product of the (R)-SA.
This problem that may arise in the course of quantitation of the enantiomeric composition of a selectand may be illustrated by a constructed example:
Effect of Enantiomeric Impurity of CDA on Quantitative Results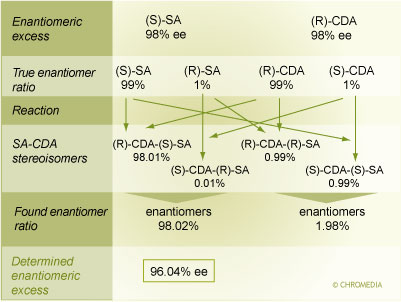
Let us consider a selectand (S)-SA with an enantiomeric excess (ee) (link to Analysis of enantiomer composition) of 98% which corresponds to a true enantiomer ratio of 99% S-enantiomer and 1% R-enantiomer. If the chiral derivatizing agent (R)-CDA is of the same enantiomeric purity it contains 1% (S)-CDA. Upon reaction, 4 stereoisomers will be formed. If we assume for simplicity an identical reaction rate, the 4 stereoisomers will be produced in a ratio of 98.01% (R)-CDA-(S)-SA, 0.01% (S)-CDA-(R)-SA, 0.99% (R)-CDA-(R)-SA, and 0.99% (S)-CDA-(S)-SA. The former two and the latter two isomers are enantiomeric to each other and will be co-eluted on the RP-HPLC system. Hence, a peak area ratio and thus enantiomer ratio of 98.02% (S-enantiomer) and 1.98% (R-enantiomer) would be determined. This corresponds to an enantiomeric excess of 96.04% which is obviously incorrect.
Consequently, it is of utmost importance to know the enantiomeric impurity content in the chiral derivatizing agent. This must be determined experimentally (note: do not rely on the specifications of the supplier). This can be done by validation with an independent method (preferentially a direct approach) or by analysis of a selectand with known validated enantiomeric excess. If the enantiomeric excess of the CDA is known, the systematic error that would be made by not considering the enantiomeric impurity in the CDA can be compensated for by appropriate correction factors.
- No racemization, either of selectand or chiral derivatizing agent, must occur. This can be assured by derivatizing and analyzing an enantiomeric or non-racemic selectand with known enantiomer composition.
- No kinetic racemate resolution must occur.
Such a phenomenon may be observed if the rate constants for the derivatization reaction are significantly different for the R- and S-enantiomers i.e. kR ≠ kS.
For example, let us consider a case where we derivatize a racemate and the rate constant of the R-enantiomer is higher than that of the S-enantiomer kR > kS. If the reaction is stopped before completion, the analyzed enantiomer ratio would deviate from the 50:50 ratio as expected for a racemate e.g. (R)/(S) = 55:45. This phenomenon is called kinetic racemate resolution and would make a significant systematic error. Hence, it must be avoided by completion of the reaction (i.e. increase of the reaction time). This is also supported by addition of the CDA in a large excess, e.g. 10-fold molar excess or more.
Kinetic Racemate Resolution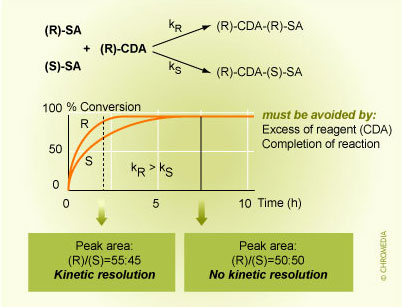
-
Another issue to be considered in the course of analysis of enantiomeric compositions by indirect methods is related to detection.
One should bear in mind that in the case of indirect enantiomer separation methods diastereomers are detected. Of course, diastereomers may differ in their detector response. For example, the extinction coefficients for the individual diastereomers corresponding to the peaks of R- and S-enantiomers in UV detection schemes may deviate significantly from each other (typically by a factor of 1.1 and 1.2). Likewise, fluorescence intensities and ionization efficiencies in mass spectrometric detection may, for diastereomers, differ considerably as well. If the enantiomer composition is supposed to be determined via the area percentages of the peaks corresponding to the two diastereomers, the ratio of the response factors of the diastereomers has to be considered as a correction factor.
 Did you ever try to explain separation to your employees or students? Well, try no more: Lee Polite did it for you in a way which is hard to beat. We will open up one example of his whiteboard class.
Did you ever try to explain separation to your employees or students? Well, try no more: Lee Polite did it for you in a way which is hard to beat. We will open up one example of his whiteboard class. 




