Abstract Gas Chromatography is a very complex and sensitive technique and is therefore sensitive to disturbances and has a high risk to have trouble. The demands on the instruments and columns are high in terms of sensitivity, accuracy, reproducibility. Trouble shooting is therefore a part of our job. To become a good – fast and efficient - trouble shooter, one must understand the separation itself as well as the instrument. A chaotic search for the problem can result in damaging the instrument, breaking or replacing parts without a plan and an wrong interpretation of the observed problems.
This chapter will help you to interpret the observations and bring structure in your troubleshooting efforts.
LevelBasic
In general:
- Check (with a checklist) the entire GC system
- Do either simple checks or test analyses
- Use an injection control standard
- Look at all available information in the chromatogram
- Compare results with other (standard) injections
So before you get out your wrenches and screwdrivers, start pushing buttons, turning controls, replace parts or call your boss or the service department, check:
- The electrical connections. Is everything plugged in? Do you have "Volts"?
- The gas supply: do you have pressure? Pressure valves open?
- Check all gasflows: column gas velocity, check split flow, septum purge, detector gasses, make-up gas
- Are there any leakages (use a leak detector instrument)?
- Check the temperature of all systems
- Is the correct method used? Are all settings correct?
- Column type right? Is the column correctly installed?
- Check the (auto) injector
- Syringe OK?
- Check the sample /check the standards, type and date
- Check the sample pre(treatment
- Check detector signal, height and stability
Extract all relevant information from a testchromatogram, check for "ghost"peaks and baseline disturbances. We advise to use a blank or a clean testsample.
Observe the following items, ![]() be precise:
be precise:
- Solvent peak (t0)
- Retention times –Absolute –Relative to standard peak or t0
- Peak shape (symmetry, tailing, width, height,
shoulders) - Separation / resolution of all peaks
- Number of peaks
- Ghost peaks
- Base line Stability –level –noise / wander
- Peak size (relative area or height)
Observe the peak shape:
- Do you see any fronting or tailing of peaks?
- Are all peak
 asymmetrical? If not, is there a relation between the chemistry of the component and the peak shape?
asymmetrical? If not, is there a relation between the chemistry of the component and the peak shape? - Tailing of certain high-polar components can indicate active spots (adsorption) in the system or damage of the injector insert.
- If all peak are 'split' there is a large chance that injection was not fast
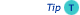 enough (mainly with manual injection)
enough (mainly with manual injection)
Here are to examples to show how you could approach.
We have two chromatograms. The chromatogram on the left is the expected one. The chromatogram on the right is not what we want.
1. Observe the two chromatograms.
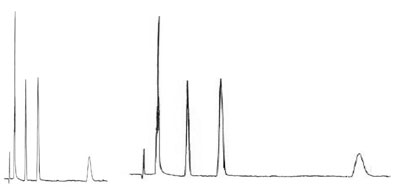
2. Collect your observations.
Here we see:
- A shift in all retention times
- No shift in relative retentions
- N has improved
- Generally no change in asymmetry
- Resolution increase?
3. Analyse your observations.
4. Make possible explanations and draw preliminary conclusions
5. Action: Test these conclusions e.g. by changing the relevant parameters
Another example of an unexpected shift in retention times:
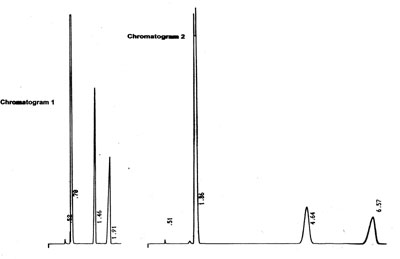
What are our observations?
- A shift in retention times
- No shift in relative retentions
- N has improved
- Generally no change in As
- Have all peaks shifted? Do we see an
 unretained component?
unretained component?
Here we see two chromatograms of a blank temperature run from 0 - 300 0C. The bottom one is the original blank run, the top one after some time of use of the system.
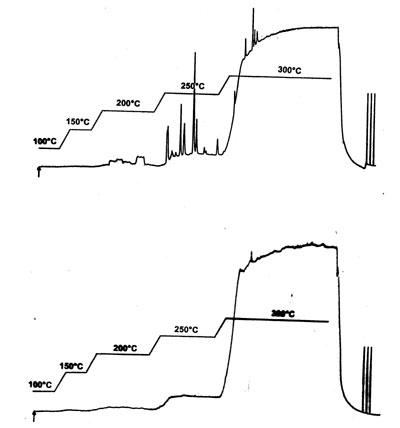
- The top one shows a number of sharp peaks (no sample injection!)
- The bottom one shows a normal run, with increasing level of the baseline at temperatures over 200 0C.
- The bottom chromatogram shows an increasing baseline with normal and expected bleeding pattern of the column.
- Bleeding and noise at the high temperature the top chromatogram is not bigger than the blanc reference.
So what could this be?
The quality of the column seems to be no problem since bleeding and noise appear OK.
- The peaks in the top one could be remainings from an earlier injection, however the peaks are quite sharp (while the older injections would give wider peaks) which makes us think this could be some contamination at or near the entrance of the column.
- A repetition of a blank analysis or just with a clean solvent injection, can prove this.
- Cleaning the injector (and maybe cutting some 20-30 cm from the inlet of the column) will most likely solve this problem. We advice therefore to remove several inches (6-10) of the column in case of poor blancs.
 Did you ever try to explain separation to your employees or students? Well, try no more: Lee Polite did it for you in a way which is hard to beat. We will open up one example of his whiteboard class.
Did you ever try to explain separation to your employees or students? Well, try no more: Lee Polite did it for you in a way which is hard to beat. We will open up one example of his whiteboard class. 




