About the chromatogram generator
Jeroen Billen, GE Water and Process Technologies, BelgiumAbstract Is investing many thousands of euro's or dollars worth the gain in time? The chromatogram generator can help you with this and many other optimization questions. Gert Desmet and Jeroen Billen with help from Peter Schoenmakers created and upgraded this tool. The file can be downloaded in Resolution Topic Circle - where you will find a number of video lessons also -. It is explained in an interview with Gert Desmet and Jeroen Billen.
LevelBasic
What can you do with it?
You can do a virtual optimization of an existing reference separation, only requiring the variation of the experimental parameters that are readily available to the experimentalist, such as pressure, temperature, column type, and length. An important feature in the output of the program is that apart from the parameters that characterize the quality of a separation, such as plate number, retention time, resolution and peak capacity, chromatograms are generated as well. Any chromatographer with the least bit of experience is familiar with this type of output and can directly make a visual verification whether or not the modified chromatographic conditions will suit the needs.
How did this start?
At the occasion of the HTC-10 meeting (Bruges, February 2008) together with Prof. Peter Schoenmakers we were asked to entertain a discussion session about the wealth of choice chromatographers currently have if they want to purchase a new instrument or column type. Should one, for example, opt for an instrument equipped with a pump that is able to deliver higher pressures or instead invest in a more standard, cheaper HPLC-instrument that is coupled to an high temperature oven? In other words: what are the effects of pressure and temperature on the required analysis time and separation efficiency. The developed tool readily allowed showing the effects of these two different instrument upgrades on a given separation.
To make the discussion session more entertaining, the discussion session was set in a casino theme. The audience was invited to play a game of casinography, wherein the goal was to achieve the fastest or most qualitative separation with only a limited budget to spend on instruments, ovens and columns.
Does this mean that it is a gimmick or is it more than that?
Although it was presented in a somewhat humorous and playful framework during the HTC-10 conference and on a later date at a meeting of The Royal Society of Chemistry (London, April 2008), the tool has a solid scientific background and improving a given separation turned out to be a challenging exercise in some cases, even for the most experienced chromatographers amongst the attendees. They enjoyed playing and saw that making predictions is harder than it looks.
Is it easy to use?
The tool was built so that the user is not required to fully comprehend or interfere in the underlying calculations in order to experiment with the tool. The program input is very simple, it allows for a manipulation of the pressure, temperature, column type, particle size and column length. These are all variables that are readily available to every chromatographist.
We want to encourage people from a beginner level on to work/play with the tool. The only requisite to fully appreciate the tool is that the user is able to analyze a chromatogram and knows how to interpret the characterization parameters such as analysis time, plate number, isocratic peak capacity and resolution.
Who should use it?
We see it as an educational tool. Therefore anybody teaching chromatography, and this can be at a university as well as at an in-company workshop or tutorial, can use it as an illustrative gizmo for his or her students to clarify the effects of different parameters on the separation speed and efficiency. Allowing the students to play with the parameters themselves will give them a better insight on the parameters influencing a chromatographic separation. In general, anybody curious to find out what the different effects of different instrument upgrades are on the goodness of a separation are encouraged to use it. However, it will also help practitioners to understand the effects of parameter changes better.
How does it work, can you explain it in a bit more detail?
The starting point of the optimization exercise is the separation of a mixture of seven components and a t0-marker on a packed bed column of 4.6x250 mm packed with 3.5 µm particles. With the application of 400 bar and the column set at 30°C, the separation is completed in just under 23 minutes (cfr. chromatogram in Fig. 1a). The components were chosen in such way that 3 pairs of components form critical pairs of which the resolution is poor (Rs < 2).
The next step is to improve this reference separation. If a complete base-line separation is desired, the quality of the reference separation is insufficient. The aim would therefore be to improve the efficiency without further prolonging the analysis time. On the other hand, for some analyses base-line separations are not the primary requirement and the reference separation meets the needed resolution. The aim would then be to enhance the speed of analysis while maintaining the given resolution.
The experimental parameters that can be varied to do this are a) the maximal deliverable pressure of the pump, b) the temperature of the oven, c) the type of column, d) the size of the particles or the domain size of the monolith that column is packed with and e) the length of the column. As for the column type, choice can be made between a typical silica packed bed column, a silica monolith column or a porous shell column.
The effects of changing one or a combination of parameters can be not only be seen visually by the changes in the chromatogram, but also in terms of series of well-known characterization parameters as retention time, retention factor of the last eluting component, achievable number of plates, or plate height, isocratic peak capacity and the resolution of the three critical pairs (table 1).
|
| Reference system | Improved system 1 | Improved system 2 |
| dp (µm) | 3.5 | 1.8 | 3.5 |
| Dptot (bar) | 400 | 1000 | 400 |
| T (°C) | 30 | 30 | 65 |
| Lcol (mm) | 250 | 150 | 300 |
| dcol (mm) | 4.6 | 4.6 | 4.6 |
|
|
|
|
|
| k’max (/) | 10 | 10 | 9.17 |
| Ntot (/) | 33,000 | 38,900 | 31,300 |
| tR,tot (min) | 23 | 11.6 | 14.9 |
| Rs (/) | 0.995 | 1.09 | 0.99 |
| np (/) | 111 | 121 | 119 |
|
|
|
|
|
Could you give us an example?
1a. Starting point. Can we get a faster separation? 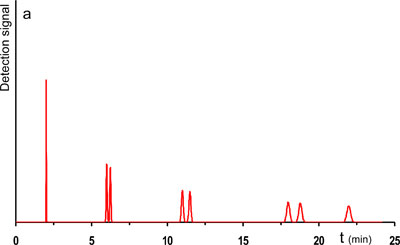
Let us set our goal at improving the speed of the reference separation, without a loss in resolution and peak capacity. Two different strategies can be envisioned. A first strategy involves an increase of the pressure drop. If the pressure drop over the system would be increased and the same column would be used, and hereby I mean maintaining the length and particle size from the column of the reference system, the separation would indeed be faster. However, these higher flow rates have a drawback as well. In the reference system, ran at 400 bar, the flow rate corresponded more or less to the optimal mobile phase velocity. This is the velocity that matches the minimal plate height in a van Deemter curve. In short, band broadening was minimized for that system. Now, using higher pressures leads to a shift of the mobile phase velocities away from the optimum of the van Deemter curve, into the region of higher plate heights, most often referred to as the C-term dominated range of velocities. The loss in resolution and peak capacity in this setup would be unacceptable. The solution is to be found in switching to columns packed with smaller particles, going from 3.5 µm to particles smaller than 2 µm. Because smaller particle diameters lead to lower plate height values the same number of plates can be obtained by shorter column lengths. Due to a lower permeability of these columns however, higher inlet pressures are required to achieve the optimal velocity (corresponding to the minimal plate height value). Following this strategy, the pressure was increased from 400 to 1000 bar and the 3.5 µm column, 250 mm in length was replaced by a 150 mm column packed with 1.8 µm particles and the separation could be achieved in half the time. Looking at the resolution and peak capacity it can be seen that these even improved slightly. The applied input parameters and resulting output are shown in table 1 (improved system 1) and the chromatogram of the improved separation is shown in Fig. 1b.
1b. Improved, now with smaller particles.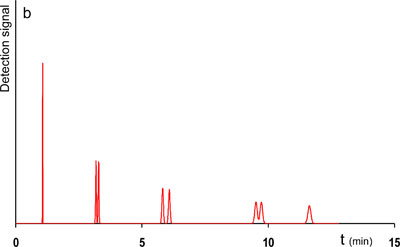
An alternative strategy could be to keep your original 400 bar pump, but switch to higher temperatures. Due to the decrease of the solvent viscosity with increasing temperature, higher mobile phase velocities can be achieved with the same pressure drop over the column and thus reducing the retention time. In addition, the molecular diffusivity of the solutes increases, which leads to lower plate height values. However, from a given temperature on the k’-values start to diminish which consequently has a negative effect on the resolution and peak capacity. It should be noted here that the mobile phase composition is changed automatically in the program, hereby striving to maintain the retention factor (k’) of the last eluting component, being k’max = 10. Of course there are limitations to the mobile phase composition adjustment and when these are reached a drop in k’ is unavoidable. For this example, let us increase the temperature from 30°C to 65°C. This only marginally influences the k’max-value and the resolution and peak capacity could be maintained by making the column slightly longer, from 250 to 300 mm. As a result the analysis time was reduced by 33%. The input and output parameters are shown in table 1 (improved system 2) and the chromatogram is visualized in Fig. 1c.
1c. Improved system 2. Now with higher temperature.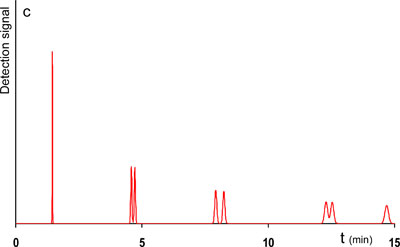
Hereby it is demonstrated that both approaches lead to a significant reduction of analysis time. The elevated pressure approach seems to be the best of both, but requires purchasing an instrument that is able to deliver pressures up to 1000 bar and the use of columns packed with sub 2 µm particles. It will therefore roughly cost € 20,000 – 25,000 (calculated for a time span of 3 years) more than the elevated temperature option. The consumer should than decide if the extra gain of just over 3 minutes per separation is worth this amount of money.
The tool was created in Microsoft Excel. Won’t this limit the number of people that can use it?
It is indeed true that the tool is an Excel template containing all underlying mathematical relations between the physical and adsorption parameters and the separation efficiency. Our choice for Excel was largely based on its ease of use, its simplicity to combine a numerical and graphical output and the speed at which a presentable and useful tool could be made. After all, at first the Excel template was only intended for that one discussion session at the HTC conference. It was only afterwards, when people asked for a copy to experiment with themselves, that it became clear to us that the template had a lot of potential.
Providing the tool as an Excel template still means that we can reach a lot of people with it, as Excel is probably the most commonly used spreadsheet program. Nevertheless, we are considering to develop a more professional tool for the future. However, we haven’t decided on how this exactly should look like. One possibility is to go to an online separation calculator, or another option is to make a stand-alone executable.
The most surprising result?
What was to you the most surprising result when using the tool?
An important factor of the tool that hasn’t been mentioned is that it accounts for extra column band broadening as well. This means that axial dispersion coming from the injector, connection tubing and detector are incorporated in the calculation of the efficiency. One of the most striking results when testing the tool was the impact of the contribution these extra-column sources of band broadening can have on the total band broadening of the system. This is especially true for high speed separations, where short columns packed with small particles are used. Extra-column band broadening therefore certainly needs to be accounted for.
How does the development of such a tool fit in the research activities of your research group?
The department of chemical engineering of the Vrije Universiteit Brussel focuses its research activities to a large extent on the unravelling and understanding of the different sources of band broadening. After all, this information is vital in order to purposefully enhance the separations of tomorrow. Our group, which consists of nine PhD students and two post doctoral researchers, approaches this topic from an experimental as well as from a theoretical side, supported by computational studies. One interesting output was the distribution of the kinetic plot program, which allowed attributing the kinetic performance of a given chromatographic system and comparing it with other systems or columns in an unbiased way.
You could say that the optimization tool is a sort of junior version of the kinetic plot program. The amount of information you can get out of it is less, but the output is easier to interpret.
Do you have any plans for the future with this tool?
Apart from updating it from an Excel template to a more professional program, we have been thinking about several other options to enhance the tool for the future. One is that cost of optimization is re-introduced in the program. This would however require a more rigorous enquiry on the prize people have to pay for their instruments and columns.
Another thing is to provide updates of performance data every time a new type of column has become commercially available. Or, a version of the template could be made available in which the performance data are set as variables, in order to give users the option to fill in their own van Deemter data. This would, however, have as a consequence that the introduction level of the tool becomes higher. Therefore a possibility would be to provide two versions. The first one would be an easy-to-use version with a restricted number of changeable parameters, hereby mainly aiming at educational purposes. This version would be similar to the one that is available now as up till now this wide variety of changeable parameters was left out, because this might scare people to try the tool instead of winning their interest. Next to this basic version a Pro-version of the tool can be made in which far more parameters are taken into account. For example, momentarily, the supplementary band broadening coming from the instrument, is fixed. In the extended version, variables such as tubing length and diameter and injector and detector volumes could be added. This would give the experimentalist an initial idea if it would be beneficial to invest in the reduction of the extra-column band broadening and if so, how much in terms of efficiency could be gained.
